COVID-19 vaccine might be ready in a few months. Given India’s leadership position in vaccine manufacturing, it is likely that the bulk of these vaccines would be produced in India. But developing and manufacturing vaccine is only a part of the solution. In 2021, a massive vaccine distribution program will be underway, and at that time, one of the most difficult questions the world will have to answer would be “how to distribute these vaccines safely to every village of the world?”. Since vaccines are highly temperature sensitive, ensuring that they reach every country’s health distribution system in the right condition would be a considerable challenge. Studies estimate that over 25% vaccines are wasted every year for cold chain issues, which we can ill afford when it comes to COVID-19.
Even today as India speeds up its testing capabilities for COVID-19, it is transporting samples and test kits over thousands of kilometres to make them reach an approved lab. These samples and kits can get spoilt at any point during this journey, if compromised for temperature. This makes the entire fight against COVID-19 vulnerable.
The truth is temperature-controlled supply chain is highly complex and fragmented. There are multiple stake holders and intermediaries through the entire process and a single miss can result in damaged products. At Pluss, we took upon this challenge and asked ourselves that as experts in temperature control, can we address this problem reliably, economically and sustainably? This marked the genesis of India’s first Phase Change Material (PCM) based validated temperature-controlled packaging- Celsure®.

PCMs are advanced materials that can store and release large amount of energy keeping the temperature constant without any external power supply. This is achieved by energy involved in change of phase from solid to liquid or vice versa. The most commonly available such material is water/ice (gel). However, its efficacy as PCM has always been in question. Melting at 0°C (32°F), use of water exposes the vaccines to the risk of freezing, potentially compromising them.
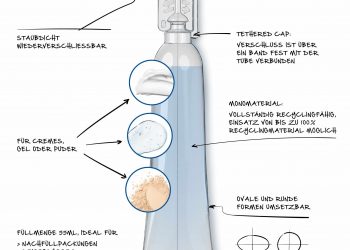
Using our expertise in PCM technology and a patent pending design, we developed a range of pre-qualified pharmaceutical shipping boxes that maintain the temperature of vaccines or other heat labile products for up to 120 hours between 2-8 deg C, 15-25 deg C and frozen (-20 deg C) as the case may be, with a variety of payload volumes (https://www.pluss.co.in/celsure/). Besides the precise temperature control and prolonged back up hours, the unique design of the solution allows for five-minute assembling without the thawing step that usually takes an hour.

Priced affordably, Celsure® has managed to bridge the gap that existed between either using a low cost un-validated shipping box or the imported, more expensive validated, PCM based shippers. Developed in India, Celsure® is also validated as per the standards set by International Safe Transit Association (ISTA).
A good temperature-controlled packaging is the one that just keeps the required temperature- neither less nor more. In doing that, Celsure® ensures that users get the inside temperature as asked for. Now one can ship between Mumbai to Moscow without worrying much about the ambient.

M.D., Pluss Advanced Technologies
Samit has an M.Sc. in Physics and Bachelor in Electrical and Electronics Engineering, from the Birla Institute of Technology & Science, Pilani, India (1994). He also holds an M.S.in Electrical Engineering. from the University of Hawai’i, USA (1997). As MD at Pluss, Samit heads the overall strategy, marketing and finance portfolios. Pluss is committed to create value through products and services designed to benefit people and create a better world.
With a belief that Energy access is going to be the key for a sustainable world, he has led the growth of energy storage materials and their applications at Pluss. A decade of work in smart materials allowed Pluss to become the leading player in enabling thermal energy storage products. He led his team to develop and create products – some of which were the first in the world – MiraCradle™, worlds first neonatal cooler can keep a baby cool for 3 days. This device has become the standard of care to treat birth asphyxiated babies in India. The challenge of moving pharmaceuticals and vaccines at the right temperature, was addressed through – Celsure™. Pluss went on to receive the CII’s – Most innovative MSME award twice – In 2015 and 2017. He has spoken at several national and international conferences over the years on promoting energy storage.
Extremely passionate about Environment, he engages in Environment conservation through Advit Foundation, a Gurgaon, India based NGO. An avid marathoner, Samit has successfully completed the Los Angeles Marathon, the Delhi Half Marathon and the Mumbai Marathon.
Vice President- Business Development, Pluss Advanced Technologies
Udit is a management post-graduate specializing in Marketing & Finance from Management Development Institute (MDI) Gurgaon and has a Bachelors in Technology in Biotechnology from Amity University, Noida, India.
Udit has over 11 years of experience in the area of life sciences and healthcare. Before joining at Pluss, he has held different roles ranging from product management to business management in several companies in the life science industry including in Bio-Rad Laboratories and Thermo Fisher Scientific. Udit now leads healthcare and agriculture business verticals at Pluss - from strategizing the new product development to identifying business growth opportunities. He has won several awards in business management and product development and has been a speaker at several public and private conferences. One of the latest innovations he is working on is an affordable, electricity free infant warmer for low birth weight new born babies supported by BIRAC and CMC Vellore.